
Back Ammoniumsulfaat Afrikaans كبريتات الأمونيوم Arabic Sulfatu d'amoniu AST Ammonium sulfat Azerbaijani آمونیوم سولفات AZB Амониев сулфат Bulgarian অ্যামোনিয়াম সালফেট Bengali/Bangla Sulfat d'amoni Catalan Síran amonný Czech Svovlsur ammoniak Danish

| |
| |
| Names | |
|---|---|
| IUPAC name
Ammonium sulfate
| |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.029.076 |
| EC Number |
|
| E number | E517 (acidity regulators, ...) |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
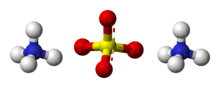
| (NH4)2SO4 | |
| Molar mass | 132.14 g/mol |
| Appearance | Fine white hygroscopic granules or crystals |
| Density | 1.77 g/cm3 |
| Melting point | 235 to 280 °C (455 to 536 °F; 508 to 553 K) (decomposes) |
| 70.6 g per 100 g water (0 °C) 74.4 g per 100 g water (20 °C) 103.8 g per 100 g water (100 °C)[1] | |
| Solubility | Insoluble in acetone, alcohol and ether |
| −67.0×10−6 cm3/mol | |
| 79.2% (30 °C) | |
| Structure | |
| orthorhombic | |
| Hazards | |
| GHS labelling: | |
 
| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
2840 mg/kg, rat (oral) |
| Related compounds | |
Other anions
|
Ammonium thiosulfate Ammonium sulfite Ammonium bisulfate Ammonium persulfate |
Other cations
|
Sodium sulfate Potassium sulfate |
Related compounds
|
Ammonium iron(II) sulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ammonium sulfate (American English and international scientific usage; ammonium sulphate in British English); (NH4)2SO4, is an inorganic salt with a number of commercial uses. The most common use is as a soil fertilizer. It contains 21% nitrogen and 24% sulfur.
- ^ Lide, David R., ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, FL: CRC Press. ISBN 0-8493-0487-3.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search
